The specific heat of hydrochloride at constant pressure is 21.4 joules per Kelvin mole, according to Georgia State University. However, specific heat for gases is expressed in constant volume as well, which means that the specific heat for HCl also is expressed as 2.57 molar heat capacity in constant volume. If it is a sodium hydroxide solution, you can assume the heat capacity is equal to that of water (=4.2. Specific Heat capacity of solid NaOH?
Depends on the concentration of the solution. Most widely usedconcentration, i.e. 48% has Cp ~ 3.331 kJ/Kg.K
What is the specific heat capacity of NaOH?
Specific heat of NaOH?
NaOH, or sodium hydroxide has a molar mass of 39.9971 grams per mole. This lends to its specific heat capacity of 59.66 Joules per mole per Kelvin. Read More
What is the specific heat of NaOH and HCl?
How To calculate Specific Heat of A compound Such as NaOH?
Specific heat is the measure of energy it takes to raise a unit mass in temperature by one degree Celsius. When measuring a compound that is water soluble, heat it separately to a specific range, then use the liquid to calculate the amount of heat that was used. Read More
What is the heat of neutralization between ch3cooh plus naoh?
Is heat generated with NaOH plus HNO3 reaction?
Are the heat of reaction between 50 ml HCl and 50 ml NaOH the same with 100ml HCl and at 100ml NaOH?
No. The heat of reaction for 50mL of each will be multiplied by 2 for 100mL of each since heat of reaction is really on a per mole product basis, and there will be twice as many moles of both HCl and NaOH in 100mL as in 50mL. Read More
What is heat of neutralization between HCl and NaOH?
How do the specific heat of the samples compare with the specific heat of water?
specific heats of the samples compare with the specific heat of water Read More
Why specific heat is large specific volume?
Specific heat has nothing to do with specific volume. Read More
Specific heat and latent heat of LPG?
What is the accepted value for the heat of solution of sodium hydroxide?
The accepted value for the heat of solution of NaOH is -44.2 kJ/mol Read More
Specific heat of rice?
What is the specific heat of sinter?
What is specific heat?
Specific heat capacity (quite often called specific heat) is the amount of heat that a given mass of a material must absorb to raise its temperature by a specific amount. Read More
Is sulfuric acid soluble NaOH?
Sulfuric acid reacts violently with NaOH, producing sodium sulfate and water and lots of heat! Read More
What is the relation between Heat capacity and specific heat?
specific heat is directly proportional to heat capacity Read More
Effect of temperature on specific heat of material?
The effect of temperature on specific heat of material is referred to as specific heat capacity. Read More
What can you say about the specific heat of sand compared to the specific heat of water?
Specific heat of sand is less than that of water. Read More
What can NaOH do to water?
eat people In water NaOH becomes a strong base and can dissolve things like human skin and is used as a drain cleaner. When small amounts of water are added to dry NaOH, the reaction generates heat. Read More
Where can you get a specific gravity conversion table for measuring NaOH into ml?
What is the specific heat of copper at STP?
What is the specific heat of Basalt?
How much heat is released when 3.600 mol naoh is dissolved in water?
How is molar heat capacity related to specific heat?
molar heat capacity is the product of specific heat capacity and molecular weight molar heat capacity=specific heat capacity*molecular weight Read More
The specific heat of a substance depends upon?
The specific heat of a substance depends on mass and temperature. ex. the specific heat of a heated pool is greater than the specific heat of a hot cup of tea. Read More
What is a valid unit for specific heat?
Specific heat capacity (not 'specific heat') is measured in joules per kilogram kelvin (J/kg.K). Read More
Which compound has the highest specific heat?
Which compound has the highest specific heat? Read More
Does water have specific heat?
Yes, all substances have a specific heat. Read More
What is the specific heat capacity by mechanical method?
Is the specific heat of water high or low?
The specific heat of water is high. An example of an object with low specific heat would be a metal pan. Since specific heat is the energy needed to raise 1g of something 1 degree Celsius, water would have a high specific heat. Read More
What is The amount of energy required to raise the temperature of one KG of a substance by one KG is determined by its?
By its specific heat. By its specific heat. By its specific heat. By its specific heat. Read More
What is the specific heat constant of oxygen?
heat constant = mass * specific heat capacity * temperature change Read More
If a substance heats up more quickly than another substance does it have a higher specific heat or lower specific heat?
What substance does not evolve heat when dissolved in water Na2SO4 NaOH CaO NaCl?
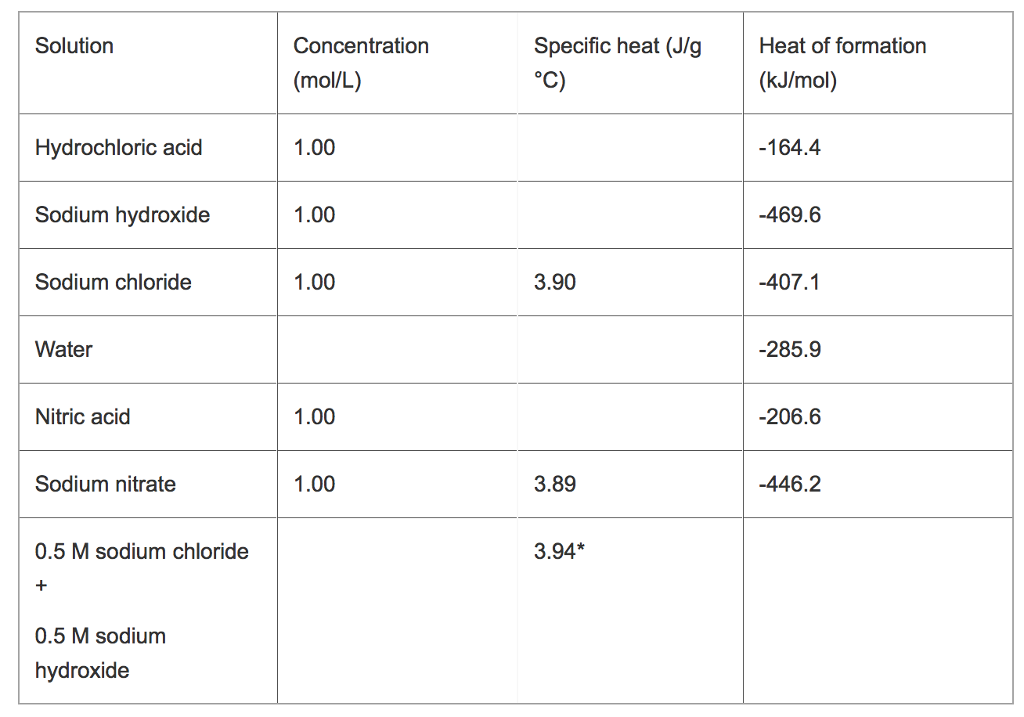
What is the specific heat of soil?
The specific heat of soil is 123457 degress celcious. Read More
Do metals have a high specific heat?
No. Metals have a relatively low specific heat. Read More
What is the specific heat of americium?
The specific heat of americium is: 0,11 J/gK Read More
What is the specific heat capacity of granite in its liquid state?
What is the specific heat capacity of Polyester?
The specific heat capacity of polyester is 2.35degrees Read More
The specific heat of which metal is 0.16?
The solid with a specific heat of 0.16 is Carborundum. Read More
Why heat evolve at dilution of caustic?
Caustic dissolution involves the dissolution of NaOH in water. The heat evolves during this process because it is being absorbed. Read More
Does water have one of the highest specific heat capacities?
With a specific heat of 1 calorie/gram, water has the highest specific heat of any common substance; ammonia is an example of a substance that has a specific heat that is slightly higher than that of water. Read More
Most specific heat capacities are found by combining a substance with another with a known specific heat usually water. But How do chemists know the specific heat capacity of water in the first place?
Examples of specific heat 1.people eat their hobos at a specific heat at 901 degree Read More
What is heat combustion?
Which of the following would be the best material to use for making tea kettles A copper specific heat 380 B iron specific heat 450 C graphite specific heat 710 D aluminum specific heat 920?
What has higher specific heat water or lead?
Water has much higher specific heat than lead. All metals have fairly low specific heat values. Read More
What substances that heat and cool quickly have a high or low specific heat?
A substance that has a low specific heat needs less heat to increase its temperature. In other words under a constant heat flux it will heat or cool more quickly than the higher specific heat substances. Read More
Specific heat of water?
The specific heat of water is 4.186 joule/gram 째C. Read More
What is the specific heat of cotton?
The specific heat of cotton is 0.072, and it is also a good insulator. Read More
